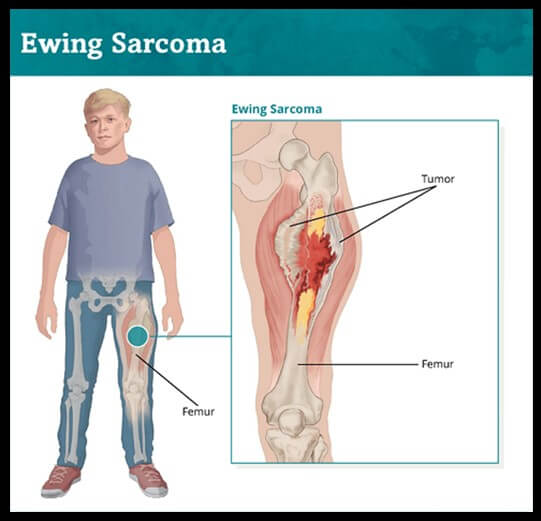
Working with a team of cancer researchers and physicians who run the Ewing Sarcoma Program at the UCLA Jonsson Comprehensive Cancer Center, we have created our first cancer drug candidate – a single-disease specific immunotherapy targeting Ewing sarcoma, a rare but deadly bone and soft tissue cancer primarily affecting children and young adults. Based on the positive data, we plan to launch FDA IND enabling studies needed to apply for approval to proceed with human trials. We look forward to the day when treating cancer will be as simple as getting a flu shot – a better way to treat cancer.
We are developing a novel Universal Cancer Vaccine (UCV), to be delivered as a shot, that uses cutting-edge bioengineering and molecular technologies to uniquely detect, mark and kill only cancer cells. By forcing cancer cells to express a unique marker that healthy cells do not have, custom antibody drugs and the body’s immune system can target and destroy them with precision. Additionally, we intended to make cancer cell express protein markers associated with known diseases such as COVID, tetanus or measles. By doing so, we hope to leverage the body’s immune system to easily destroy cancer cells as if they were previous vaccinated common diseases. We have filed a patent application for the UCV and are working with the UCLA Jonsson Comprehensive Cancer Center to develop the platform, which may be administered as a one or two-shot treatment.
Based on the foundational nanoparticle technology developed for UCV, we are developing a novel Universal CAR-T Cell therapy, to be delivered as a shot, that can reprogram natural immune T-cells inside the body to seek and destroy any targeted cancer cells.
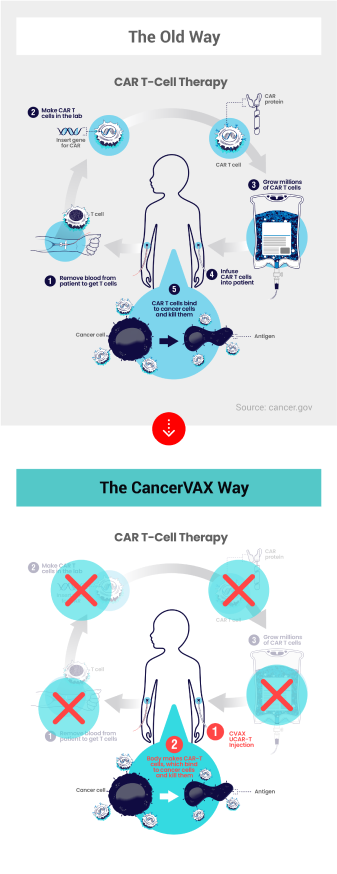
CAR-T cell therapy is a type of FDA-approved immunotherapy in which a patient’s T-cells are genetically modified in the laboratory so they will bind to specific proteins (antigens) found on cancer cells and kill them. These modified T-cells are called Chimeric Antigen Receptor (CAR) T Cells. Conventional CAR-T Cell therapy can cost more than $500,000 per dose due to complicated and expensive lab processes. We believe we can dramatically lower the cost to a few thousand dollars per dose by simply helping the body make its own CAR T-cells.